
A Sulfur-Based Battery design developed by international research teams could significantly increase how much energy a rechargeable battery can store, scientists say. The technology, reported in recent peer-reviewed studies and laboratory demonstrations in 2025–2026, aims to replace conventional lithium-ion cells in electric vehicles and renewable energy storage systems by using abundant sulfur instead of costly metals.
Table of Contents
New Sulfur-Based Battery Design
| Key Fact | Detail / Statistic |
|---|---|
| Energy density potential | Up to 2–3× higher than lithium-ion (theoretical) |
| Cost advantage | Sulfur is an industrial byproduct and widely available |
| Main obstacle | Polysulfide “shuttle effect” reduces battery lifespan |
What the Sulfur-Based Battery Means
The Sulfur-Based Battery is a rechargeable power cell that uses sulfur as the cathode material rather than the metal oxides used in lithium-ion batteries.
Modern electronics, electric cars, and grid storage rely heavily on lithium-ion batteries. These cells have improved steadily since their commercial introduction in the 1990s, but many scientists believe they are nearing performance limits.
“Lithium-ion technology is highly refined, but its energy density increases are becoming incremental,” said Dr. Linda Nazar, a battery chemist whose research has focused on lithium-sulfur battery chemistry.
Sulfur offers a unique advantage: it is light, inexpensive, and widely available. Oil refineries produce vast quantities as a byproduct of fuel processing, making supply far less constrained than lithium, nickel, or cobalt.
Why Scientists Are Interested
Higher energy density
Energy density determines how long a device runs or how far a vehicle travels before charging.
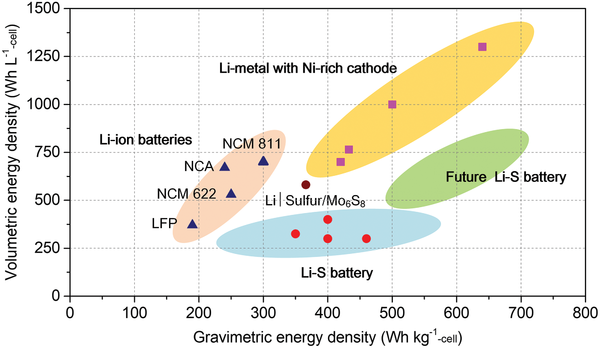
Commercial lithium-ion batteries typically deliver about 250–300 watt-hours per kilogram. Laboratory sulfur batteries can theoretically exceed 600 watt-hours per kilogram.
That could transform transportation.
An electric vehicle using lighter electric vehicle batteries could travel significantly farther on a single charge, or manufacturers could reduce battery size and cost.
Aircraft and heavy trucks, where weight matters more than size, may benefit even more.
Cheaper materials
Sulfur costs dramatically less than battery-grade lithium compounds and avoids cobalt and nickel mining concerns.
Energy economists say storage prices must drop substantially to enable reliable renewable power grids.
Lower-cost energy storage technology could help utilities store solar electricity generated during the day and use it at night.
The Problem That Blocked Sulfur Batteries
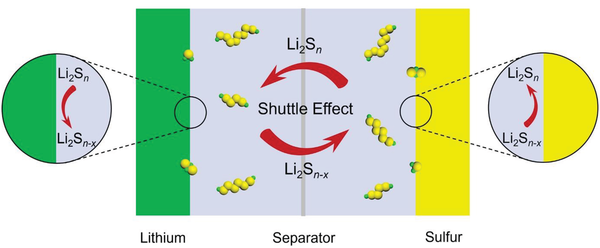
For decades, the lithium-sulfur battery failed to reach commercialization because of a chemical instability known as the polysulfide shuttle effect.
During charging and discharging:
- Sulfur turns into intermediate liquid compounds called polysulfides.
- These dissolve in the electrolyte.
- They migrate across the battery.
This causes rapid capacity loss and short battery life.
“Instead of holding charge, the battery effectively leaks energy internally,” explained Dr. Shirley Meng, a materials scientist specializing in advanced battery systems.
What the New Design Changes
Recent research combines several engineering solutions.
1. Protective separators
Researchers created nanostructured membranes that trap sulfur compounds and prevent migration.
2. Catalytic cathodes
New carbon-based and metal-doped cathodes accelerate reactions and improve stability.
3. Alternative electrolytes
Some teams are testing solid-state or gel electrolytes to prevent chemical dissolution.
Prototype cells now survive hundreds of cycles — a critical improvement toward commercialization.
Environmental Impact
The environmental implications may be as significant as the technological ones.
Traditional battery production requires mining lithium, cobalt, and nickel. These processes consume water and energy and can create pollution.
Sulfur, by contrast, already exists in large stockpiles as a petroleum industry byproduct.
Using sulfur could:
- reduce mining pressure
- lower manufacturing emissions
- simplify recycling
Researchers say sulfur batteries may also be easier to recycle because they contain fewer heavy metals.
Implications for Electric Vehicles and Renewable Energy
Electric vehicles
Range anxiety remains a major barrier to adoption. A higher-capacity battery could extend range beyond 800 kilometers without increasing weight.
Manufacturers could also lower prices because batteries make up roughly 30–40% of an electric car’s cost.
Renewable power storage
Solar and wind generation fluctuates hourly and seasonally. Utilities need long-duration storage.
Sulfur batteries could enable multi-day electricity storage — something lithium-ion systems struggle to provide economically.
Industry Competition
The Sulfur-Based Battery is not the only candidate replacing lithium-ion.
Competing technologies include:
- solid-state batteries
- sodium-ion batteries
- metal-air batteries
Each has advantages. Sodium-ion is cheaper, while solid-state offers safety improvements. Experts say the future may involve multiple battery chemistries serving different applications.
Safety Considerations
Lithium-ion batteries can overheat and, in rare cases, catch fire.
Sulfur batteries may be safer because they operate at lower internal stress and can use non-flammable electrolytes.
However, safety testing is ongoing. Engineers must confirm stability during crashes, extreme temperatures, and long-term use.
Manufacturing Challenges
Scaling from laboratory coin-cells to automotive battery packs is a major engineering hurdle.
Factories must address:
- electrode thickness
- uniform chemical reactions
- moisture sensitivity
Battery manufacturers must also redesign production lines originally built for lithium-ion cells.
Economic Impact
Energy analysts say next-generation batteries could influence global supply chains.
Countries lacking lithium reserves could still manufacture sulfur batteries because sulfur is globally available.
That may diversify the battery industry and reduce reliance on specific mining regions.
Governments are funding research partly for economic security, not just climate policy.
Global Context
Governments worldwide are investing billions in advanced battery research as part of electrification strategies.
Transportation, heating, and industry are moving toward electrification to reduce carbon emissions. This shift requires vast amounts of storage.
Energy agencies say battery capacity must grow several-fold by 2040 to support clean energy targets.
Development Timeline
Researchers describe a likely commercialization path:
2026–2028: pilot manufacturing and specialized uses
2028–2032: aviation, drones, heavy transport
After 2032: consumer electric vehicles and grid deployment
These estimates depend on cycle life and reliability improvements.
Expert Perspectives
Scientists remain cautiously optimistic.
Supporters say the chemistry solves key cost and energy barriers. Skeptics note many promising batteries fail during industrial scaling.
“The science is compelling,” one engineering researcher said at an international battery conference. “The real challenge now is manufacturing.”
Outlook
Scientists say commercialization could still take several years, but progress is accelerating.
If durability and manufacturing challenges are resolved, sulfur batteries may become a major component of future power systems. As one materials scientist noted in a research overview, “The chemistry has long promised transformative storage — now engineering must prove it.”
FAQs About New Sulfur-Based Battery Design
What is a Sulfur-Based Battery?
A rechargeable battery that uses sulfur as the main cathode material instead of metal oxides used in lithium-ion batteries.
Is it better than lithium-ion?
Potentially yes in energy capacity and cost, but durability is still under development.
When will it reach consumers?
Likely within the next decade if engineering challenges are solved.
Will phones use it?
Eventually possible, but vehicles and grid storage will likely adopt it first.
















